Blockbuster products presents huge growth potential
Scroll down to read the impact of the termination of the agreement with Pfizer
Biocon Ltd.: Company Highlights
Biocon Ltd. – The largest Biotechnology Company in India (by revenues)
Market View of Biocon Ltd (as of 12/01/2012)
Latest Stock Price: Rs. 264.25
Latest Market Cap: Rs. 5536 Cr. (Mid Cap Stock)
52 Week High Stock Price: Rs. 396.00
52 Week Low Stock Price: Rs. 240.10
Latest P/E: 15.49
Latest P/BV: 2.63
Tell me more about Biocon Ltd…
Biocon, which started its operations in a garage, is today India’s largest biotechnology company. It manufactures innovative biopharmaceuticals which cater primarily to high growth areas of diabetology, nephrology, cardiology and oncology. It also provides custom research and clinical research services through its subsidiaries Syngene and Clinigene respectively. Thus, its presence across the three critical stages of drug development – drug discovery, development & manufacturing and commercialization make it an integrated healthcare company.
The graphs given below show the revenue segregation of Biocon according to geography and different business segments.
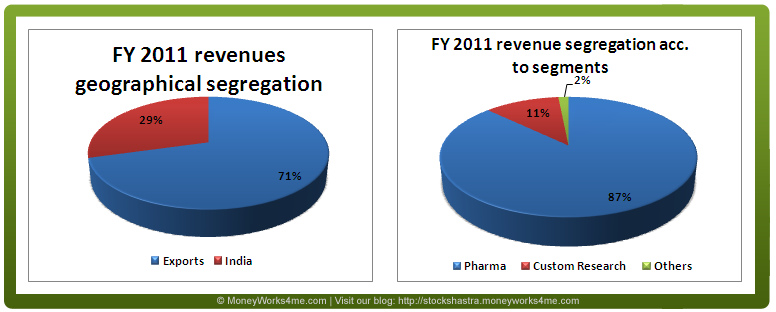
So, what does Biocon exactly do?
Biocon’s business spans across 3 basic divisions as follows:
1. Biopharmaceuticals: These are medical drugs produced using fermentation technology and synthetic chemistry skills. They include the following products:

2. Custom Research: Carried out through Syngene, a 99.99% owned subsidiary of Biocon, this involves research on and creation of new drugs for MNC Pharma and Biotech companies.
3. Clinical Research Services: Carried out through Clinigene, a 100% owned subsidiary of Biocon, this involves carrying out clinical trials (Phase I, II, and III) for testing the safety and efficacy of medicinal drugs.
How has the financial performance of Biocon Ltd. been? Here’s the review…
Considering the significant contribution of its subsidiaries, the analysis of Biocon’s financial performance has been done on a consolidated basis.
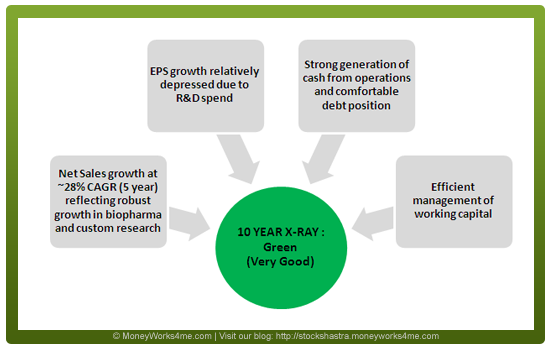
The10 YEAR X-RAY of Biocon Ltd. shows that the performance of the company has been quite good since the company went public in 2004.
Biocon’s consolidated Net Sales has registered a CAGR of ~28.5% driven both by biopharmaceuticals (5 Year CAGR of 32%) and custom research services (5 Year CAGR of 28%). The growth in biopharmaceuticals has largely been driven by its statins portfolio which commands over 20% global market share.
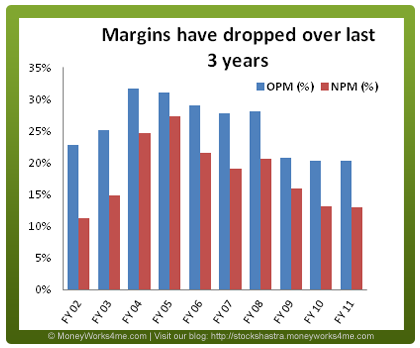 Its operating and net profit margins were high during the period of 2004-2008 but thereafter have decreased over the last 3 years. As a result EPS growth has been subdued as compared to Net Sales growth with a 5 year CAGR of 15.5%. Over the last 6 years, Biocon has spent around 6% of its Net Turnover on Research & Development on an average. The company operates in the field of Biotechnology where the gestation period for the products is quite long. As a result, it takes a long time to get products from R&D phase to market. To add to this, successful development of a product leading to market launch is not guaranteed. This partly explains the relatively low growth seen in Biocon’s Net Sales and EPS as compared to other Pharma companies.
Its operating and net profit margins were high during the period of 2004-2008 but thereafter have decreased over the last 3 years. As a result EPS growth has been subdued as compared to Net Sales growth with a 5 year CAGR of 15.5%. Over the last 6 years, Biocon has spent around 6% of its Net Turnover on Research & Development on an average. The company operates in the field of Biotechnology where the gestation period for the products is quite long. As a result, it takes a long time to get products from R&D phase to market. To add to this, successful development of a product leading to market launch is not guaranteed. This partly explains the relatively low growth seen in Biocon’s Net Sales and EPS as compared to other Pharma companies.
Book Value per share has shown good growth; the company has also been generating consistently positive and high cash flow from operations. Return on Invested Capital which indicates management’s efficiency of utilizing funds has also been good with the 6 year average figure at 17.4%.
The debt position of the company is very comfortable with the Debt to Net Profit figure and Debt to Equity figure standing at 0.95 and 0.17 respectively for FY 2011. Infact, as of September 2011, the consolidated debt of Biocon stood at Rs. 332 Cr. whereas cash on books stood at Rs. 318 Cr. making it a virtually net debt free company. Working capital management has also been good and receivable days have decreased (61 as of FY 2011) over the last 10 years and inventory days (50 as of FY 2011) have been consistent.
Thus, while Biocon’s financial performance has been pretty good; its balance sheet reflects a strong and comfortable position making it a safe company as far as financial performance is considered. Considering this, we can say that Biocon’s 10 YEAR X-RAY is Green (Very Good).
What can we expect in the future? Here is the fundamental analysis of Biocon Ltd.
In the short term
 Profitability expected to stay muted due to R&D investments:
Profitability expected to stay muted due to R&D investments:
Biocon has entered into several strategic partnerships with pharma companies like Pfizer and Mylan (Read detail in long term prospects). In these partnerships, Biocon has reached a stage wherein it will have to invest significant resources into the development of the biogeneric products which will result in increase in R&D expenses. While a part of the licensing revenues has already been recognized, commercialisation of biogenerics will take at least 2 years and hence profitability is expected to be muted due to mismatch between revenues and costs.
Sales of Fidoxamicin API to drive growth:
The US FDA has approved Optimer Pharma’s Fidaxomicin drug (an antibiotic) for which Biocon is the sole API supplier owing to its partnership with Optimer. Fidaxomicin has been claimed to have fewer side-effects and a more targeted treatment than existing treatments like Vancomycin and Metronidazole. Vancomycin generates ~USD250m sales in the US and a similar amount outside the US and Optimer will have to get a share from this market. However, there has been better-than-expected off-take in Fidaxomicin API and Biocon is focused on improving operational efficiencies to meet the increased demand.
Thus, considering all these factors, we can say that the Short Term Future Prospects of Biocon Ltd. are Orange (Somewhat Good).
In the long term
Blockbuster drugs in the pipeline:
Biocon has developed an oral insulin research molecule IN-105 which is in its Phase III clinical trials. The company had earlier planned to launch it first in India and then globally. However, it then changed its strategy and is now planning to go for a global launch by licensing the product to an MNC pharma company for which it has been scouting. If Biocon successfully out-licenses this NCE, it is likely to lead to a significant re-rating as the product has the potential to eventually replace injectable insulin in future. IN-105 is thus a global blockbuster market opportunity for Biocon.
Another blockbuster opportunity that Biocon has is its research molecule T1h or Anti-CD6 monoclonal antibody which is currently undergoing Phase III clinical trials in India. T1h is effective in the treatment of Psoriasis and Rheumatoid Arthritis (RA) and has a potential target market of over USD 20 billion by 2015. The company has similar plans to out-license this molecule to an MNC pharma or Biotech Company. Few drugs are present in these therapeutic segments and hence it presents good growth opportunities for Biocon.
Research and Marketing partnerships the way forward:
Over the last few years Biocon has struck research and marketing partnerships with a number of players in a bid to access the global markets and this is expected to be an important growth driver for the company in the future. After a molecule/drug being developed by Biocon reaches the clinical trial stage (usually proof of concept), Biocon looks to find a global partner to out-license the molecule and share the development costs. In lieu of out-licensing the molecule, Biocon gets licensing income in the form of milestone payments after specific steps/results in the development process are achieved. The partner then helps commercialise the products in the global markets owing to its marketing muscle and shares some part of the profits with Biocon. This model de-risks Biocon to a certain extent and also enables it to focus on its strength i.e. R&D without worrying much about the commercialization aspects.
The table given below lists some of the major partnerships that Biocon has struck.
As most of the patented biotechnology drugs are going off-patent from 2015 and the global pool of innovative drugs are facing a cliff, global MNCs have no option but to look at biosimilars to protect their top line. This presents a great opportunity for Biocon.
Research Services to grow rapidly in the future:
Biocon’s subsidiaries Syngene (custom research) and Clinigene (clinical research) are likely to report strong growth of over 30% over the next few years due to an increase in outsourcing by global pharma and biotech companies. Syngene’s client base consists of more than 60 companies including seven of the top ten MNC companies. One of its biggest clients is Bristol Myers Squibb (BMS) with more than 400 scientist working on BMS projects. Biocon has also now moved to providing more value added and high-end research services which has reflected in higher growth in the research services division.
Focus on emerging countries:
The global pharmaceutical industry is shifting course for growth and profitability. Tier 3 countries like Venezuela, Poland, Argentina, Vietnam etc. have joined the original emerging countries like India, China, Brazil, Russia etc. These countries offer strong growth prospects fuelled by their rising GDPs, increasing access to healthcare and improving regulatory environment. In FY 2011, Biocon registered a growth of close to 40% in the emerging markets business. The current emerging market estimate for the biosimilar insulin business is about US$ 1.5 billion with a 5-year CAGR of 15%. This market is estimated to grow to US$ 5 billion by 2020. Biocon is looking to increase its footprint in all emerging markets through strategic alliances. Early investments in R&D and manufacturing have given Biocon the advantage of cost-competitive products within key segments. This can help it grab a large market share in these emerging markets.
So, after seeing the company’s positive points, is there anything you should be concerned about?
- Heavy dependence on statins business: Biocon is heavily dependent on its statins business which faces high competition from China. Moreover, to remain cost competitive, it has to continuously upgrade technology. A new treatment in this therapeutic category may also impact sales and profitability of the company.
- High risk from top customers: The Company faces huge risk to its revenue profile if it loses any of its major customers. As it supplies API to the generic manufacturers, the loss of regulatory approval might lead to loss of revenues and profits. Biocon has major dependence on Bristol Myers Squibb (BMS) – a large customer for recombinant human insulin and custom research from Syngene.
- Competition from Chinese players: Biocon faces tough competition from the Chinese players. It also faces competition from US and European companies who have huge installed capacities and greater financial strength. As far as custom research business is concerned, it faces competition from other CRAMS players in India like GVK biosciences, Jubilant Life Sciences, Advinus Therapeutics etc.
Despite these concerns, Biocon is well-positioned to make the most of the opportunities in the biologics space. Hence, we can expect the long-term future of the company to be Green (Very Good).

So, is it an investment-worthy company?
Biocon is the largest biotech company in India. It is an innovation and research driven company with a strong biopharmaceutical portfolio. Its research and marketing deals are expected to drive growth going forward. More such deals are expected to follow with blockbuster products in its pipeline. This combined with its high-end research services and increasing focus on emerging countries will drive its growth in the future.
Yes, Biocon Ltd. is an investment worthy company, but only at the right price. Being a mid-cap stock (Market cap of Rs. 5536 Cr.), Biocon is considered to have relatively high risk as compared to large cap stock.
Currently, its stock price is at Rs. 262.25 (as on 12 Jan’ 2012). Though the company has been beaten down in the current negative sentiment, it is very important to check if this current price offers an attractive discount to its right value (MRP) or is it over-priced? It is always best to invest at an attractive discount to its MRP, to get maximum returns at minimum risk. Become a member of MoneyWorks4me.com to know its sensible buy- price and hence take the right action for this company.
Apart from buying at a discount to the MRP, investors can also look at the Technical chart of a company to support their buying and selling decisions based on fundamentals.
So, let’s see what the technical chart of the company indicates? Click to view the chart
This chart of Biocon indicates few important traits about the stock. The stock, after correcting from its pre-crash-all-time high of Rs 325 in Nov 2007 and reaching all time low in Dec 2008, rallied substantially and made a new all time high at Rs 477 in Oct 2010 (unlike majority of the stocks that could not even cross their pre-crash high!). After touching the new high, the stock has been trading in a downward channel for a year but has only retraced 61.8% (till now) of the previous rally. 61.8% is a major psychological support level for any stock. A bounce back has also been observed from this level. Incidentally, this Fibo level is also converging with important channel support line. Though the stock is still in downward trend till the channel is broken, it will be safe to assume that the downside will be limited from here. From a technical perspective, investors could consider accumulating in small quantities around Rs 240-260 levels with a stop-loss of Rs 215.
However, remember technicals can be best utilized if used as a supporting tool to fundamentals. So, make sure you check out the MRP of Biocon Ltd. at MoneyWorks4me.com
Update – Termination of the deal with Pfizer: What’s the impact on Biocon?
In October, 2010, Biocon signed a mega-marketing deal with Pfizer, the world’s largest Pharma Company, for worldwide commercialization of four bio-similar insulin products. The market for these products was estimated to be around $14 billion (Rs. 62,300 crore). According to the deal, Pfizer was to pay Biocon $200 million (Rs.900 Cr.) upfront, half of which was kept in an escrow, to be released over two years. Pfizer was also to pay an additional sum of $150 million over a longer period, subject to completion of milestone events apart from royalty payments on the subsequent sale of these products.
The deal gave Pfizer the worldwide rights to market four of Biocon’s key insulin products—glargine, aspart, lispro and recombinant human insulin. The deal was beneficial to Biocon as it lowered its risk from launch of products, minimized investments and working capital requirements while giving it access to Pfizer’s huge marketing network.
However, on 12th of March, Pfizer called off this deal and both companies agreed to cancel the deal stating that “both companies have decided to move forward independently owing to individual priorities for their respective bio-similar businesses”.
So, how does this impact Biocon?
As part of the settlement, Biocon will keep all the upfront and milestone payments received from Pfizer and no refund will be made. Pfizer will pay an additional amount to Biocon towards the settlement of this particular deal; this amount was not disclosed by the company. According to some reports, this amount could be in the range of $150-200 milllion. The payments will be addressed and will be reflected in Biocon’s fourth-quarter earnings. (to be announced in May 2012)
All rights licensed to Pfizer would revert to Biocon as of March 12, and all insulin distributed under the brand name UniviaTM and GlarviaTM would be commercially available from Biocon Ltd only, and would be exclusively manufactured, supplied, marketed and supported by Biocon.
As far as impact on performance of Biocon is considered, the deal will not have any short term implications as such because the development proceeds would have been spread over a period of 5 years.
However, long term growth will definitely be impacted to a certain extent, as the deal also included recurring royalty payment from sale of insulin products in the future. The market has reacted negatively to this happening with the stock price falling by more than 8% in the day. The major concern here is that Insulin market is dominated by only three players — Novo Nordisk, Sanofi and Eli Lilly and is tough to crack. This may have also prompted Pfizer to walk away from selling Insulin products.
We feel that while Biocon will be able to market these products in emerging markets, penetration of regulated markets will remain a big concern. Forging strategic alliances with global pharma players is also going to be difficult as Pfizer is the largest player in the world and its reluctance to continue to deal reflects the challenges in the market. It may have found it difficult to crack the US and Europe markets (which account for 70-80% of the total insulin market), and if that is the case, Biocon or any other company will surely find it very difficult to penetrate these markets.
Biocon in a statement, however, said that it will continue to work with its existing partners in several markets and will pursue a commercial strategy on its own and through new alliances in other markets for these insulin products. It is now considering tying up with regional partners to market its insulin product basket in the different regions. How this shapes up and whether the company can rope in new partners remains to be seen.
So, what is the impact on the MRP of the company?
As mentioned above, the deal is not expected to have any significant impact on the short term financials of Biocon. In fact, the next quarter financials will see cash coming in from the severance pay that Biocon will receive from Pfizer. Biocon is expected to use these funds for construction of its bio-similar facility in Malaysia. However, it may have to raise some funds to make up for the shortfall now which earlier would have been contributed by the Pfizer deal.
The cancellation of the deal does impact the long-term growth rate of Biocon to a certain extent. It also indicates the difficulties that the company could face in its bid to crack the insulin market in US and Europe. Further, we feel it could also lead to contraction of the PE multiple commanded by the company. Biocon is currently trading at a trailing PE of 14.65 considering consolidated financials while its 5 year historical average PE has been close to 19.
Considering the cancellation of the deal, and its impact, we have reduced our EPS growth rate and PE estimates for the company which have resulted in reduction of Biocon’s MRP to a certain extent. To check the updated MRP of Biocon log on to MoneyWorks4me.com.
Concluding, while the cancellation of the deal does raise some questions over the growth potential of the company with respect to its insulin products, the fundamentals of the company are not impacted greatly. Biocon has a strong business model with some blockbuster products in the pipeline. We expect the stock price to remain depressed in the short term owing to the overhang of this event coupled with less than expected financial performance over the last few quarters. Improved performance of the company and alliances with big Pharma players for insulin and other products could act as triggers for the stock in the future.
Disclaimer: This publication has been prepared solely for information purpose and does not constitute a solicitation to any person to buy or sell a security. It does not constitute a personal recommendation or take into account the particular investment objectives, financial situations or needs of an individual client or a corporate/s or any entity/ies. The person should use his/her own judgment while taking investment decisions.










A nice short cut of Biocon to guide the investor.
glad to see very detailed analysis….with figures…